Lipoderma is a Non-Surgical Fat Restoration Solution
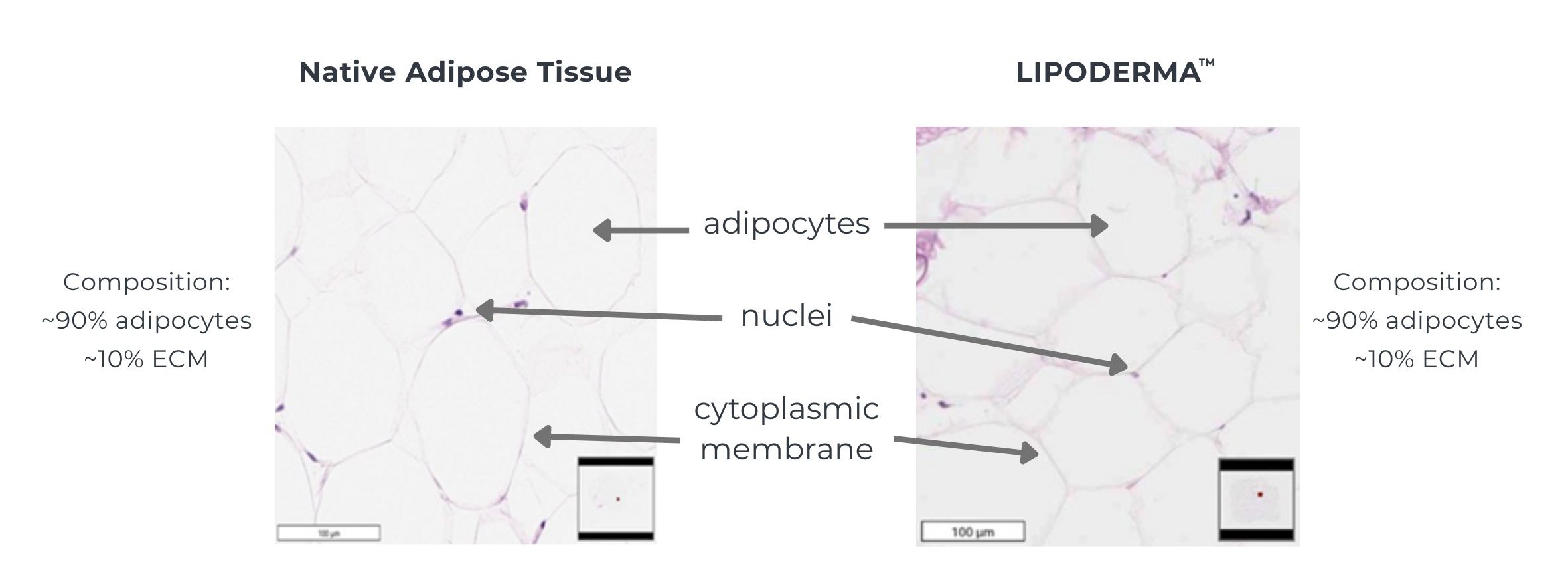
Lipoderma, a cryopreserved human adipose allograft, is aseptically processed without irradiation, removing immunogenic components while preserving the extracellular matrix, growth factors, and intact adipocytes— the essential elements for natural tissue restoration, often referred to by plastic surgeons as the ‘good stuff.’
ADIPOSE TECHNOLOGY YOU CAN TRUST
Setting a New Standard -
Lipoderma Restores the Good Fat Lost During Rapid Weight Loss and Aging
Each vial of Lipoderma is processed to preserve the native cellular components (adipocytes) and structural components (extracellular matrix – ECM) of adipose tissue while removing immunogenic components, ensuring consistency, safety, and reliable outcomes.
For more information, please refer to our Safety and Clinical Overview.
Natural Fat Restoration - Convenience Without Compromise
Unlike traditional fat grafting, Lipoderma does not require harvesting a patient’s own fat, making it an option for those with limited donor tissue or wishing to avoid additional surgical procedures. The controlled preparation also eliminates much of the variability associated with fat transfer, where results can depend heavily on harvesting and processing techniques.
Contrary to dermal fillers, Lipoderma remodels to become the patient’s own tissue, which supports longer-lasting results and a more natural outcome. Easy, comfortable, and natural, Lipoderma restores what’s yours.
If you have any questions about who might be a good candidate or what the typical procedure is like, please refer to our Q&A with Dr. Fearmonti, MD of Plastic & Reconstructive Surgery.
Frequently Asked Questions
What is Lipoderma?
Lipoderma is a human adipose tissue allograft used for the repair, reconstruction, or replacement of adipose tissue defects. It preserves the native architecture and components of fat, including the extracellular matrix and signaling molecules, to restore natural volume and support healthy tissue remodeling.
How is Lipoderma classified?
Lipoderma meets the FDA’s requirements for Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) under Section 361 of the Public Health Service Act. It is FDA-registered and manufactured in compliance with American Association of Tissue Banks (AATB) standards for donor screening, recovery, and processing.
How is Lipoderma processed?
Lipoderma is produced from adipose tissue recovered from eligible cadaveric donors. Donors are fully screened and tested in accordance with Food and Drug Administration (FDA) and American Association of Tissue Banks (AATB) requirements. The tissue is aseptically processed in a cleanroom environment and each lot is tested for sterility to ensure product safety and quality.
Does Lipoderma contain viable cells?
No. Lipoderma contains intact, but devitalized adipocytes.
Does Lipoderma revascularize?
Yes. Nonclinical studies demonstrate host-driven revascularization, with new vessel formation observed as early as three days after implantation.
What happens to the DNA within Lipoderma?
Residual DNA within devitalized adipocytes is naturally degraded after implantation. In vivo testing shows no detectable human DNA by four weeks post-implantation.
Does Lipoderma trigger an immune response?
No. The manufacturing process removes immunogenic components such as lymph nodes and blood cells while retaining the structural adipocytes. Devitalization reduces antigenicity, and both in vitro and in vivo studies confirm that Lipoderma does not elicit an immune response.
What are the safety data to date?
To date, zero product-related adverse events have been reported. Lipoderma has demonstrated an excellent safety profile in clinical use.
