VALIDATION OF AI AND TRADITIONAL TECHNOLOGIES
WE PROVIDE

Industry
Biotech | Pharmaceutical | Medical Devices | Pharma-chemical | Herbal medicines | Medicinal Gases | Sanitizing | Clinical Research | Cosmetic | Personal hygiene | Perfumes | Distribution and storage | Veterinary | Food | Farming | Clinical lab | Calibration lab | Testing lab | Equipment and systems | suppliers of these companies

What
SaaS software for all validation types (paperless) | Computer System Validation | Equipment Qualification | Utilities Qualification | IT/OT Infrastructure Qualification (including cybersecurity items) | Implementation of Integrity and Data | Governance projects | Supplier | Qualification | Project management

How
Software paperless GO!FIVE® | Resource allocation | Training and consulting | Turn-key projects (closed scope for the execution of qualifications and validations) | Audit
FIVE client has option (or not) to continue using the paperless model after the project is completed.
The validation documents are delivered in electronic format.
FIVE in Numbers
Quality and/or Innovation Awards*
Years of experience
Companies served
Executed projects
Market professionals trained by FIVE
Hours of training provided to the market
Hours of training for FIVE's internal team
Hours of training that the FIVE team received abroad
*11 SINDUSFARMA Quality Awards – SINDUSFARMA is the largest pharmaceutical industries association in Brazil, representing more than 90% of the market.
*1 CPhI PHARMA AWARDS SHORTLIST 2022, Category Regulatory Procedures & Compliance – Europe



- Time-consuming and burdensome
- Bureaucratic and inefficient
- Prone to human error and failure
- Slow to execute and update
- Costly to maintain and audit
- Interruptions in production
- Delays in product registration and market entry
- Exposure to regulatory fines
- Potential product recalls
- Negative impact on brand reputation

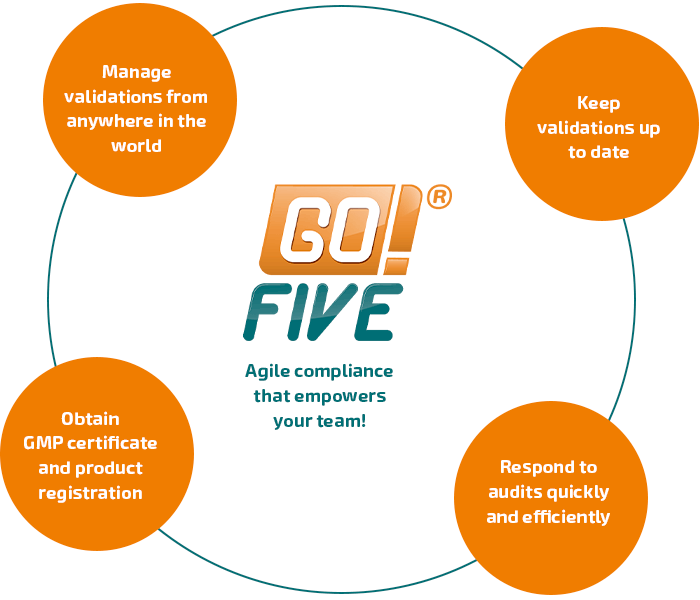
Validate with GO!FIVE®
Get more speed in product time-to-market
THE VALUE
WE GENERATE
SOME
CLIENTS
EXCLUSIVE
CONTENT
LEARN MORE ABOUT
OUR PROGRAMS

Be our Sales Rep!
If your company works for corporate clients in the Life Sciences area, already has contacts with this market to sell products or services, and understands that validation can add value to your business; maybe your company is just the one we’re looking for to be our sales rep!

Partner Client
Is your company seeking innovation and quality in the provision of its services?
FIVE Validation offers a special condition that enables our Partner clients to monetize considerably.

GO!Startup Program
A complete package that gives you access to software, training, support, and consulting hours to meet your FDA, EMA, and WHO validation compliance requirements: 7x faster. Save 75% of time and money while increasing compliance.