Teach Ionic Bonding So It Finally Clicks
Ionic bonding doesn’t have to be abstract.
Ionic bonding doesn’t have to be confusing.
Ionic bonding doesn’t have to be memorized
When students physically build compounds — balance charges, test combinations, and see structure form in real time,
Then Understanding Replaces Guessing.
Why this works:
The Ionic Bond Disc Model turns symbolic equations into something students can manipulate, test, and understand.
● Clearer lessons
● Fewer blank stares
● Stronger retention
● Faster problem solving
If you teach chemistry, this tool was built to make your job easier — and your students more confident from day one.
Why Ionic Bonding Is Harder to Teach Than It Should Be
Students are told that ionic compounds must add up to a zero charge
—but they don't know how to get to zero.
Students memorize the criss-cross method
—but don’t understand why it works.
They balance formulas mechanically
—but can’t explain charge relationships.
They skip mole ratios
—because the foundation never clicked.
You reteach it three different ways.
And still see blank stares.
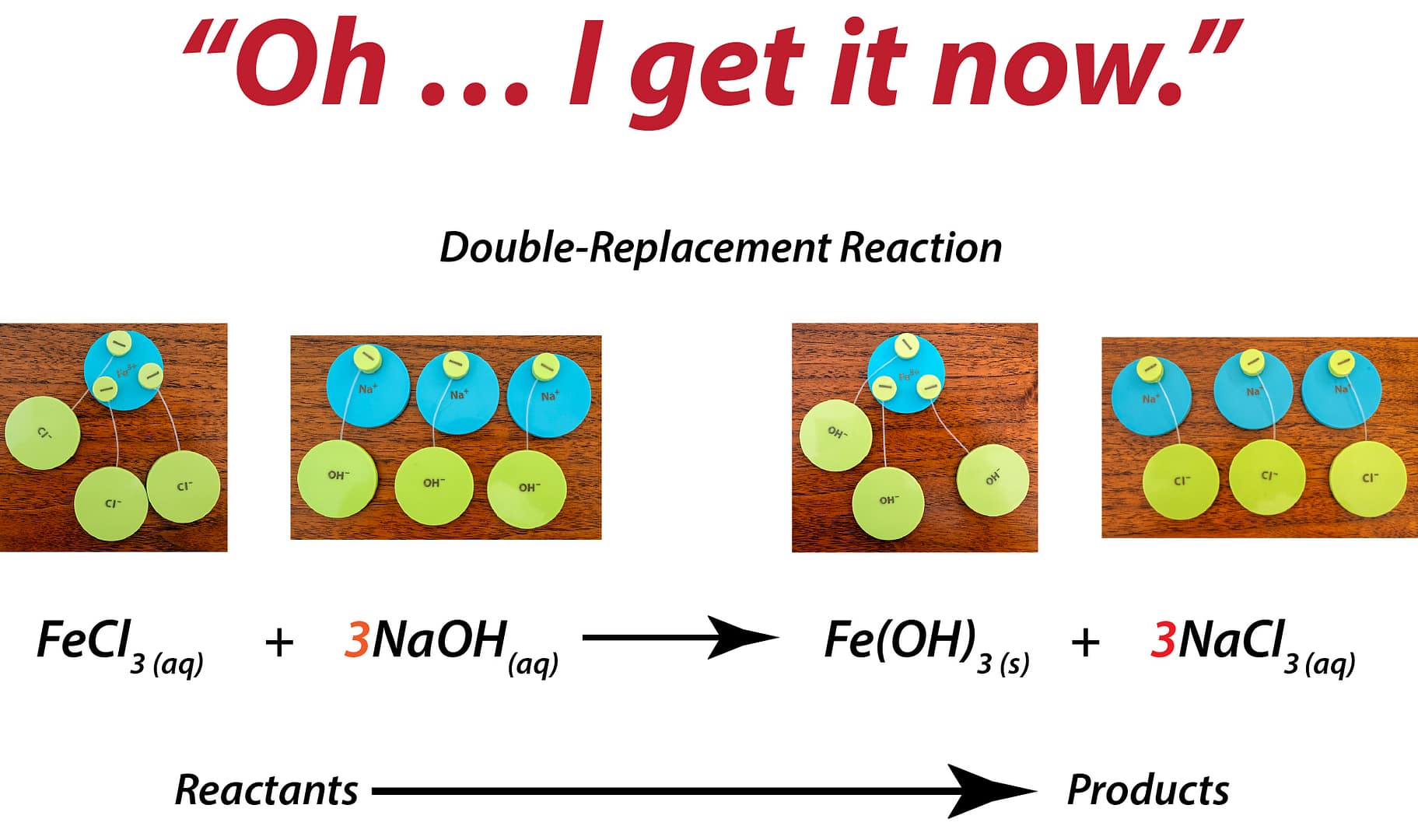
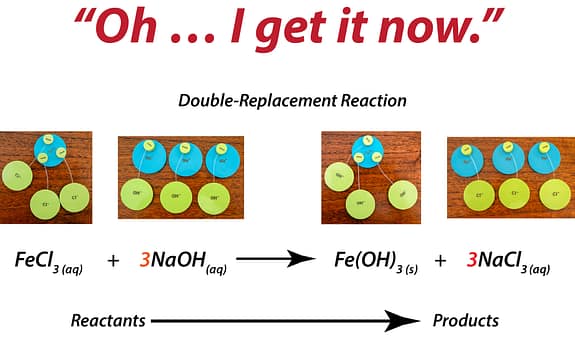
What Changes When Students Can Build It
Students don’t guess at charge balance.
They physically combine ions until the total equals zero.
The “criss-cross method” stops being a rule.
It becomes obvious.
Ratios aren’t memorized.
They’re built.
Students test combinations.
They see why some work — and others don’t.

The Ionic Bond Disc Model System
The Ionic Bond Disc Model is a structured, expandable system — from foundational classroom sets to broader ion coverage for deeper exploration.
Start where your students are.
Expand as your curriculum demands.

Calcium Nitride — Ca₃N₂