Ion exchange chromatography (IEC) offers a precise method for enhancing separation efficiency through the exploitation of ionic interactions. By adjusting parameters like pH and ionic strength, you can greatly optimize the binding dynamics between charged biomolecules and the resin. This tailored approach not only improves resolution but also elevates sample purity ion exchange chromatography, making IEC invaluable in biopharmaceutical applications. However, understanding the nuances of resin types and their impact on overall performance is essential for maximizing results.
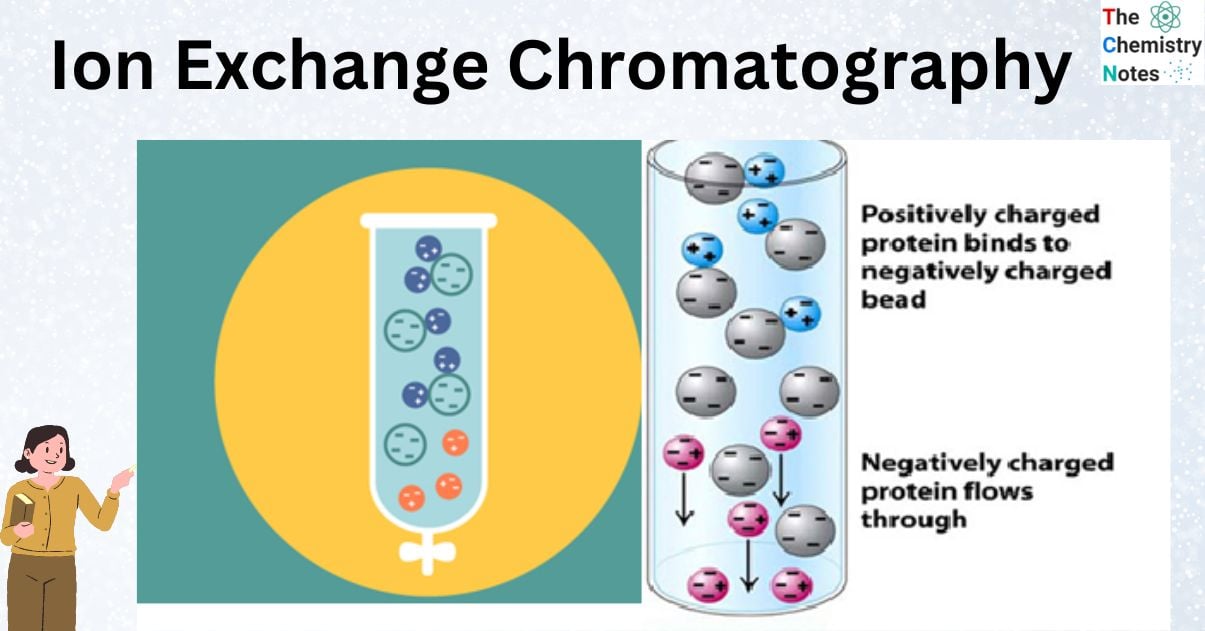
Principles of Ion Exchange Chromatography
Ion exchange chromatography (IEC) operates on the principle of electrostatic interactions between charged ions in a sample and oppositely charged groups attached to a stationary phase.
In cation exchange, positively charged ions in your sample compete for binding sites on the negatively charged resin. This competition allows you to selectively retain specific cations based on their affinity.
Conversely, in anion exchange, negatively charged ions are attracted to positively charged sites on the resin.
The efficiency of IEC depends on factors like ionic strength, pH, and the specific functional groups of the resin. By manipulating these parameters https://laballiance.com.my/, you can enhance separation efficiency and achieve high-resolution purification of target ions, making IEC a powerful technique in analytical chemistry.
Types of Ion Exchange Resins
Resins play an essential role in ion exchange chromatography, influencing the separation process’s effectiveness.
You’ll find two primary types of ion exchange resins: cation resins and anion resins. Cation resins exchange positively charged ions and are typically used for purifying metal ions or amino acids. They’re often composed of sulfonic acid groups, enhancing their selectivity for cations.
On the other hand, anion resins target negatively charged ions, making them ideal for separating nucleic acids or proteins. These resins usually feature quaternary ammonium groups that increase their affinity for anions.
Understanding the characteristics of these resins allows you to select the appropriate type for your specific application, ultimately optimizing separation efficiency in your chromatographic processes.
Factors Affecting Separation Efficiency
The choice of resin greatly impacts the separation efficiency in ion exchange chromatography.
You’ll find that the pH influence is essential; it affects the charge state of both the resin and the target ions, which can alter binding interactions. Maintaining an ideal pH guarantees that the desired ions remain in their most reactive form, enhancing their separation.
Additionally, ionic strength plays a crucial role; higher ionic strength can lead to decreased selectivity by promoting competition between ions for binding sites. Conversely, lower ionic strength can improve resolution but may require careful adjustment to avoid non-specific interactions.
Applications of Ion Exchange Chromatography
While exploring various analytical techniques, you’ll find that ion exchange chromatography plays an essential role in diverse applications across multiple fields.
In biopharmaceutical purification, it’s important for isolating proteins, enzymes, and antibodies, ensuring high purity and yield. The method effectively separates biomolecules based on charge interactions, which is crucial for developing therapeutics.
Additionally, in water treatment, ion exchange chromatography is employed to remove undesirable ions and contaminants, improving water quality for consumption and industrial use. This technique’s ability to selectively target specific ions enhances efficiency in both processes, making it an invaluable tool for scientists and engineers alike.

Advantages Over Traditional Separation Techniques
Ion exchange chromatography offers several advantages over traditional separation techniques, particularly in its efficiency and specificity.
You’ll find that these chromatography benefits considerably enhance your analytical capabilities:
- High resolution: You can achieve better separation of closely related ions, minimizing overlap.
- Scalability: The technique adapts easily from preparative to analytical scales, making it versatile for various applications.
- Rapid processing: You’ll notice quicker run times compared to other methods, enhancing throughput in your lab.
- Targeted separation: Ion exchange allows for selective removal of specific ions, improving purity in your samples.
These factors collectively contribute to a more effective and reliable analysis, positioning ion exchange chromatography as a superior choice among separation techniques.
Future Trends in Ion Exchange Technology
As advancements in materials science and engineering progress, you can expect significant innovations in ion exchange technology that will enhance its efficiency and application scope.
One notable trend is the integration of automated systems, which streamline operations and increase throughput. These systems facilitate real-time monitoring and adjustment of parameters, optimizing separation processes.
Additionally, the push towards green chemistry is driving the development of eco-friendly resins and solvents, minimizing waste and energy consumption.
You’ll also see a rise in hybrid techniques that combine ion exchange with other separation methods, increasing versatility.
As these trends evolve, ion exchange technology won’t only become more efficient but also more sustainable, making it an essential tool in various industries, including pharmaceuticals and environmental science.
Conclusion
To summarize, ion exchange chromatography stands out as a crucial technique for enhancing separation efficiency in various applications. By understanding the principles and manipulating factors like pH and ionic strength, you can achieve the best binding interactions. With specialized resins tailored for different biomolecules, this method not only improves resolution and purity but also offers advantages over traditional techniques. As advancements continue, staying informed about future trends will further empower your analytical capabilities in biopharmaceutical purification and beyond.





































