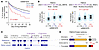Issue published March 2, 2026 Previous issue

- Volume 136, Issue 5
Go to section:
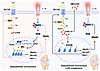
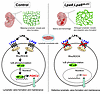
On the cover: Dual role for macrophages in muscle regeneration and degeneration
Yin et al. report that a macrophage subset expressing activin A facilitates muscle regeneration by acting on satellite cells but contributes to heterotopic ossification in pathological settings such as fibrodysplasia ossificans progressiva. The cover image shows injured skeletal muscle sections immunostained with anti–mouse embryonic myosin heavy chain (eMyHC) (magenta), marking regenerating muscle fibers; mouse IgG (light blue), indicating necrotic fibers; and Hoechst (yellow), labeling nuclei. Image credit: Wenqiang Yin, Kazuo Okamoto, and Hiroshi Takayanagi.
In-Press Preview - More
Abstract
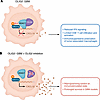
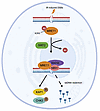
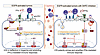
Immune evasion is a major obstacle ahead of pancreatic cancer therapy. Recent data implicate pro-inflammatory macrophages in the progression of pancreatic ductal adenocarcinoma (PDAC) and its therapeutic response. However, whether or which of the pro-inflammatory macrophage subtypes play a crucial role in the immune escape of PDAC remains unclear. Here we identify a population of CD138+ tumor-associated macrophages (TAMs), characterized by their pro-inflammatory and neutrophil-chemotactic activity, which undergo significant expansion in both PDAC patients and mouse models. These cells are elicited by a local synergy between IL-34-syndecan-1 and PGE2-EP2 signaling and are associated with immune evasion and poor clinical outcomes in patients, while also promoting immune escape and disease progression in mouse models. Mechanistically, CD138+ TAMs establish a feedforward loop with immunosuppressive Siglec-F+ neutrophils, which exhibit elevated PGE2 expression, via the secretion of Saa3 and Cxcl1. Targeting CD138+ TAMs by disrupting IL-34-syndecan-1 signaling with anti-IL-34 neutralizing antibodies significantly suppresses PDAC progression, especially when combined with anti-PD-1 antibodies. Together, our study elucidates a CD138+ TAM-Siglec-F+ neutrophil axis that drives immune escape in PDAC and proposes a therapeutic strategy that integrates IL-34-syndecan-1 signaling blockade with anti-PD-1 immunotherapy for the treatment of PDAC.
Authors
Chao Wang, Qi Zhang, Jinyan Huang, Fangyu Lin, Danyang Zhao, Youling Mu, Junshuo Tong, Jinping Li, Yingjiqiong Liang, Tao Zeng, Fukang Shi, Hang Shen, Tingting Lu, Tingbo Liang
Abstract
Lysine-specific demethylase 1 (LSD1; KDM1A) orchestrates context-dependent chromatin programs, yet its role in epithelial immunity remains largely unknown. Here, we identify LSD1 as a central brake on retinoid- and AP-1–driven enhancer activation in epidermis and demonstrate that its inhibition induces anti-tumor immunity. While epidermal LSD1 is required during development, acute loss or topical inhibition in adult skin is tolerated and triggers coordinated expression of retinoic acid signaling, lipid remodeling, and chemokine induction pathways. CUT&RUN profiling reveals that LSD1 occupies enhancer regions enriched for AP-1 motifs at retinoid metabolism, lipid homeostasis, and immune genes. LSD1 loss increases H3K4me1/2 and gene activation at these sites, licensing a poised AP-1–retinoid program. Single-cell spatial analyses show that discrete keratinocyte subsets initiate retinoid signaling to recruit dendritic cells and activate CD4+ T cell responses. Topical LSD1 inhibition suppresses cutaneous squamous cell carcinoma in two models while amplifying keratinocyte–immune crosstalk. Functional perturbations reveal that retinoid signaling partially contributes, whereas CD4+ T cells are essential for tumor control. These findings define LSD1 as a master repressor of epithelial immune competence and nominate LSD1 inhibition as a therapeutic strategy to activate retinoid–AP-1 enhancer circuits and drive CD4-dependent tumor immunity in skin cancer.
Authors
Nina Kuprasertkul, Alyssa F. Moore, Carina A. D'souza, Julia Chini, Eun-Kyung Ko, Sijia Huang, Shuo Zhang, Ashley S. Anderson, Shaun Egolf, Laura V. Pinheiro, Alison Jaccard, Claudia T. Magahis, Lydia Bao, Yann Aubert, Cyria R. Olingou, Stephen M. Prouty, Donna Brennan-Crispi, David A. Hill, John T. Seykora, Kathryn E. Wellen, Brian C. Capell
Abstract
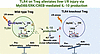
Obesity-linked steatosis is a significant risk factor for hepatocellular carcinoma (HCC); however, the molecular mechanisms underlying the transition from Metabolic dysfunction-associated steatotic liver disease (MASLD) to HCC remains unclear. We explored the role of the endoplasmic reticulum (ER)-associated protein NgBR, an essential component of the cis-prenyltransferases (cis-PTase) enzyme, in chronic liver disease. Hepatocyte-specific NgBR deletion in mice (N-LKO) intensifies triacylglycerol (TAG) accumulation, inflammatory responses, ER/oxidative stress, and fibrosis, ultimately resulting in HCC development with 100% penetrance after four months on a high-fat diet. Similarly, liver-specific knockout of DHDDS (D-LKO) NgBR’s cis-PTase partner and a knock-in model carrying a human NgBR mutation that impairs cis-PTase activity developed HCC under high-fat diet conditions, although with lower penetrance. Single cell transcriptomic atlas from affected livers provides a detailed molecular analysis of the transition from liver pathophysiology to HCC development. Mechanistically, NgBR deficiency promotes excessive hepatic TAG accumulation by enhancing lipid uptake and impairing very-low-density lipoprotein (VLDL) secretion. Importantly, pharmacological inhibition of diacylglycerol acyltransferase-2 (DGAT2), a key enzyme in TAG synthesis, abrogates diet-induced liver damage and HCC burden in N-LKO mice. Overall, our findings establish cis-PTase as a critical suppressor of MASLD-HCC conversion and suggest DGAT2 inhibition may serve as a promising therapeutic approach to delay HCC formation in advanced metabolic dysfunction-associated steatohepatitis (MASH).
Authors
Abhishek K. Singh, Balkrishna Chaube, Kathryn M. Citrin, Joseph Fowler, Sungwoon Lee, Jonatas Catarino, James Knight, Sarah C. Lowery, Sonal Shree, Keira E. Mahoney, Nabil E. Boutagy, Inmaculada Ruz-Maldonado, Kathy Harry, Marya Shanabrough, Trenton T. Ross, Stacy A. Malaker, Yajaira Suárez, Carlos Fernández-Hernando, Kariona A. Grabińska, William C. Sessa
Abstract
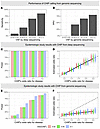
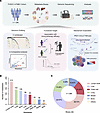
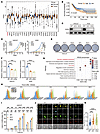
BACKGROUND. Immune checkpoint inhibitors (ICIs) targeting the programmed cell death-1 axis have revolutionized metastatic non–small cell lung cancer (mNSCLC) treatment. However, disease progression remains a concern, and the role of the complex tumor microenvironment (TME) in treatment failure is not fully understood. METHODS. In this biomarker study involving 103 patients with mNSCLC—including 81 patients who received ICI treatment—we evaluated the association between heterogeneous immune cell subsets and ICI efficacy through single-cell spatial profiling of pretreatment tumor tissue, using a 29-marker multiplex immunohistochemistry platform built for in-depth dissection of the TME. RESULTS. Among various types of intratumoral lymphocytes including T-helper 1 cells, regulatory T cells, and natural killer cells, only CD8+ T cells (TILs) were associated with ICI efficacy. Computational tissue segmentation underscored the importance of direct physical interactions between CD8+ TILs and cancer cells for ICI efficacy. TIL phenotyping identified CD39/CD103/Ki-67 positivity as a hallmark of exhausted yet functional tumor-reactive CD8+ TILs. Immunosuppressive tumor-associated macrophages (TAMs) and cancer-associated fibroblasts were independent unfavorable adversaries. High CD73 expression on cancer cells was suggested to confer tolerance to ICI in EGFR/ALK-oncogene+ NSCLC, potentially through M2-TAM accumulation and aberrant angiogenesis. CONCLUSION. Our study delineates the clinical relevance of heterogeneous immune cell subsets in ICI-treated mNSCLC, aiding the development of targeted therapeutic strategies. TRIAL REGISTRATION. Not applicable because this is a retrospective study. FUNDING. Osaka Cancer Society, KANAE Foundation for the Promotion of Medical Science, SGH Foundation, and YOKOYAMA Foundation for Clinical Pharmacology.
Authors
Kohsuke Isomoto, Koji Haratani, Takahiro Tsujikawa, Shuta Tomida, Yusuke Makutani, Masayuki Takeda, Kimio Yonesaka, Kaoru Tanaka, Tsutomu Iwasa, Kazuko Sakai, Kazuto Nishio, Akihiko Ito, Kazuhiko Nakagawa, Hidetoshi Hayashi
Abstract
Huntington’s disease (HD) is a fatal neurodegenerative disorder characterized by progressive motor dysfunction, cognitive decline, and striatal neuron degeneration, primarily affecting medium spiny neurons (MSNs). Despite extensive research, the underlying metabolic vulnerabilities contributing to HD pathogenesis remain poorly understood. In this study, we employ RNA sequencing (RNA-seq) and metabolomics analyses to identify marked dysregulation of one-carbon metabolism in HD. We validate that SHMT2, a key mitochondrial enzyme in the mitochondrial one-carbon (mt-1C) pathway, is substantially downregulated in HD patient-derived iPSC-differentiated human striatal organoids (hSOs) and YAC128 mice. Functionally, pharmacological inhibition or genetic deletion of SHMT2 exacerbates mutant huntingtin (mHTT) aggregation, induces MSN degeneration in hSOs, and impairs motor function in WT mice. Conversely, SHMT2 overexpression attenuates MSN degeneration in HD-hSOs and improves motor performance in YAC128 mice. Mechanistically, SHMT2 deficiency leads to homocysteine (HCY) accumulation, which interacts with AARS1 and suppresses histone lactylation, thereby perturbing transcriptional regulation and associating with neurodegenerative phenotypes. Finally, we demonstrate that the HD clinical drug haloperidol modulates SHMT2 expression and restores histone lactylation, providing a pharmacological tool to probe SHMT2-dependent metabolic and epigenetic regulation in HD models. These findings highlight a metabolic-epigenetic axis as a promising therapeutic target for HD.
Authors
Mingqin Lu, Kexin Li, Shanshan Wu, Zhilong Zheng, Xinyue Li, Shengda Wang, Hanwen Yu, Chunyue Liu, Yueqing Jiang, Xueqin Song, Yan Liu, Xing Guo
View more articles by topic:
Sign up for email alerts
Review Series - More
Series edited by Daniel J. Drucker
Clinical innovation and scientific progress in GLP-1 medicine
Series edited by Daniel J. Drucker
Therapies targeting the glucagon-like peptide 1 (GLP-1) receptor have revolutionized the treatment of obesity and diabetes. This series of reviews, curated by Dr. Dan Drucker, describes the latest research in this fast-moving in field, from our evolving understanding of the mechanism of GLP-1 receptor signaling to the medicines’ impact on inflammation and the consequences for heart, kidney, and brain health. The reviews also explore the impact of these medicines on conditions beyond their initial indications, including cancer and neurodegenerative disease risk.
×