Today on Drug Discovery & Development
Moving beyond the binary: how the convergence of small and large molecules is reshaping pharmaceutical R&D
Pharmaceutical R&D is at an inflection point. Consider that small molecules accounted for 65% of FDA novel drug approvals in 2025. That is a jump from 56% in both 2023 and 2024. The note that even as biologic sales are growing three times faster than their chemical counterparts and are projected to overtake them by…Lilly’s triple agonist achieves 16.8% weight loss in phase 3 trial
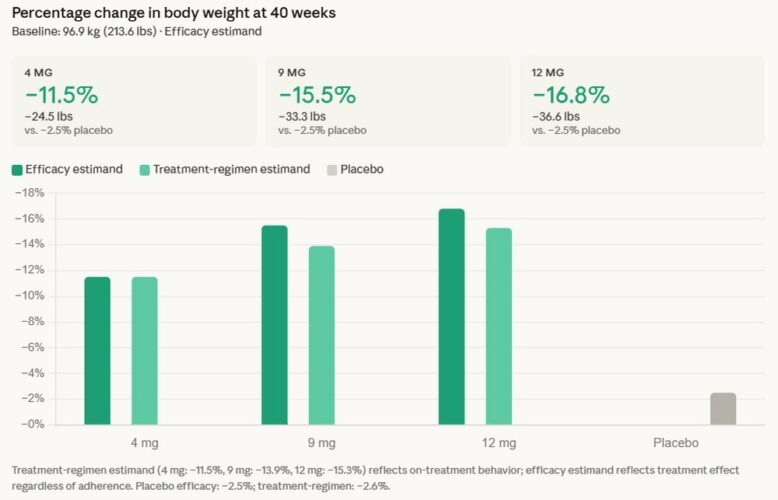
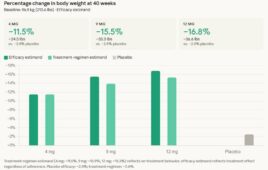
Eli Lilly’s triple agonist, retatrutide, demonstrated an average weight loss of 16.8% in a Phase 3 trial for the treatment of type 2 diabetes, the company announced today. Retatrutide is an investigational first-in-class GIP, GLP-1 and glucagon triple hormone receptor agonist. The drug met the primary and key secondary endpoints, delivering superior A1C reduction and…Drug Discovery and Development See More >

SLAS 2026: Orchestration patforms, API-first instruments and the rise of semiautonomous labs
SLAS 2026 in Boston (Feb. 7–11) drew the usual mix of liquid-handler demos and poster sessions, but this year’s show floor had something of a different energy. Three orchestration platforms launched in the same week. Multiple instruments shipped API-first. Just two days earlier, on Feb. 5, OpenAI and Ginkgo Bioworks shared results from an autonomous…
Sponsored Content See More >
Genomics/Proteomics See More >

Columbia-CZ team develops 10.3M parameter model that outperforms 100M parameter rivals on cell type classification
A new foundation model called GREmLN from a Columbia and Chan Zuckerberg Biohub team, delivers superior cell-type classification with only 10.3 million parameters, outpacing rivals like the 100-million-parameter scFoundation. Released July 9 on bioRxiv, it taps gene regulatory networks to achieve a 0.929 macro F1 score on immune cell data. “Instead of using large language…
Spatial biology: Transforming our understanding of cellular environments

Biotech in 2025: Precision medicine, smarter investments, and more emphasis on RWD in clinical trials

Genomics in 2025: How $500 whole genome sequencing could democratize genomic data

St. Jude pioneers gene editing and structural biology to advance pediatric research
Infectious Disease See More >

FDA refuses to review Moderna’s mRNA flu vaccine
Moderna received a refusal-to-file (RTF) letter from the FDA’s Center for Biologics Evaluation and Research (CBER) for its investigational mRNA vaccine for influenza, the company said Tuesday. The letter is inconsistent with the feedback received at pre-Phase 3 and pre-submission consultations, Moderna said. The company has requested a meeting with the FDA. The vaccine, mRNA-1010,…
Oncology See More >
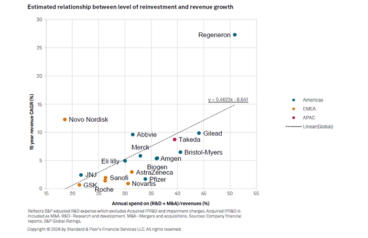
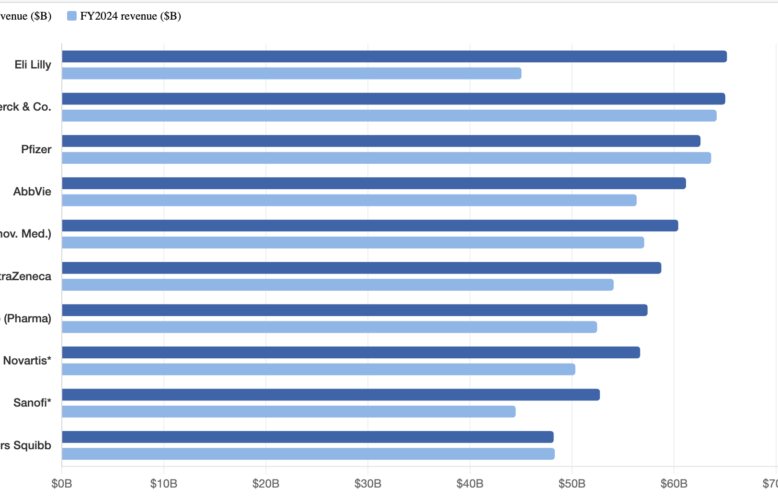
S&P report highlights Big Pharma’s concentration risk amid pre-JPM deal flurry
Merck is an A+ credit, a global oncology powerhouse, and still, about half its 2024 pharma revenue came from a single product: Keytruda, a product whose loss of exclusivity will hit around 2028 in the U.S. S&P Global Ratings calls that kind of concentration a “material weakness.” Merck isn’t alone. A new S&P analysis of…