Today on Pharmaceutical Processing
Wilmington PharmaTech invests $50M in API manufacturing expansion
Wilmington PharmaTech announced a $50 million investment to expand its Delaware campus for active pharmaceutical ingredient (API) manufacturing. Newark, Delaware-based Wilmington PharmaTech said the expansion more than doubles its current API reactor capacity. It adds two new 10,000-liter reactor suites to increase commercial-scale manufacturing capabilities. This supports growing demand for high-quality, U.S.-based manufacturing as the…Cambrex expands US, European API manufacturing footprint
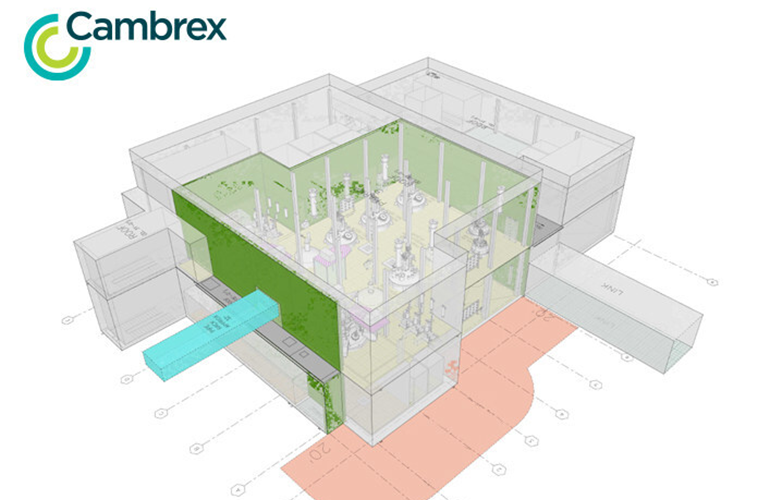
Cambrex announced today that it achieved multiple milestones in its effort to expand its active pharmaceutical ingredient (API) manufacturing. East Rutherford, New Jersey-based Cambrex said it completed the initial engineering studies for a new, large-scale API manufacturing plant in Charles City, Iowa. It also added further investment to support its Milan, Italy, site. The latest…Sponsored Content See More >
Regulatory See More >

Putting patients first in clinical trials
Clinical trials offer clinicians a unique opportunity to lead advances in medicine, granting access to cutting-edge therapies that address unmet patient needs. They are a cornerstone of clinical practice that enhance patient outcomes, particularly for those with complex or refractory cases who have exhausted standard treatment options. Clinicians understandably prioritize the well-being and interests of…

Confidently navigate the transition from bench to batch

FDA approves Merck’s Winrevair to treat pulmonary arterial hypertension

Report: Japan health authorities investigate Kobayashi Pharmaceutical factory after five deaths

Eicos Sciences’ FDA-approved Aurlumyn promises to drastically reduce frostbite amputations
Learn how to rapidly develop smart and sustainable engineering and production processes See More >
Personalized medicine: advanced technology is saving lives
The concept of a “batch size one” in pharmaceutical manufacturing is closer than ever. There are companies advancing cell therapies in which body cells are used to heal or support the individual patient. These cells are manipulated or edited to change certain biological characteristics before being transferred to the patient. Personalized medicine is truly about…
Facility See More >

MGS opens 300,000-square-foot Wisconsin facility to scale drug delivery device manufacturing
Healthcare CDMO manufacturer MGS has brought a 300,000 square foot drug delivery device manufacturing facility online in Richfield, Wisconsin. The move expands U.S. production capacity as demand for complex combination products strains available cleanroom capacity . FDA approved a record number of combination products in recent years, and the 505(b)(2) pathway has made device-drug combos…
Supply Chain See More >

Thermo Fisher, SHL Medical collab on drug delivery device production
Thermo Fisher Scientific today announced a strategic collaboration with drug delivery system provider SHL Medical. The collaboration coincides with the expansion of sterile fill-finish and auto-injector final assembly capacity at its Ridgefield, New Jersey site. Together, Thermo Fisher says its investments establish a fully integrated U.S.-based offering across sterile manufacturing, device assembly and commercial packaging…
Recalls See More >

Report: Japan health authorities investigate Kobayashi Pharmaceutical factory after five deaths
Japanese health authorities searched a Kobayashi Pharmaceutical factory in Japan following reports of five deaths related to dietary supplements, Reuters reports. The plant inspection in western Japan is the second in recent days as health authorities inspected the company’s Osaka facility. In addition to the five reported deaths, there have been more than 100 hospitalizations…